What is matter?
Matter is anything that occupies space and has rest mass (or invariant mass). It is a general term for the substance of which all physical objects consist. Typically, matter includes atoms and other particles which have mass. Mass is said by some to be the amount of matter in an object and volume is the amount of space occupied by an object, but this definition confuses mass and matter, which are not the same.Different fields use the term in different and sometimes incompatible ways; there is no single agreed scientific meaning of the word "matter," even though the term "mass" is better-defined.
Phases of Matter
States of matter are the distinct forms that different phases of matter take on. Three states of matter are known in everyday experience: solid, liquid, and gas. Other states are possible; in scientific work, the plasma state is important. Further states are possible but do not normally occur in our environment: Bose-Einstein condensates, neutron stars. Other states, such as quark-gluon plasmas, are believed to be possible. Much of the baryonic matter of the universe is in the form of hot plasma, both as rarefied interstellar medium and as dense stars.
Historically, the distinction is made based on qualitative differences in bulk properties. Solid is the state in which matter maintains a fixed volume and shape; liquid is the state in which matter maintains a volume which can vary only slightly, but adapts to the shape of its container; and gas is the state in which matter expands to occupy whatever volume is available.
Historically, the distinction is made based on qualitative differences in bulk properties. Solid is the state in which matter maintains a fixed volume and shape; liquid is the state in which matter maintains a volume which can vary only slightly, but adapts to the shape of its container; and gas is the state in which matter expands to occupy whatever volume is available.
|
|
|
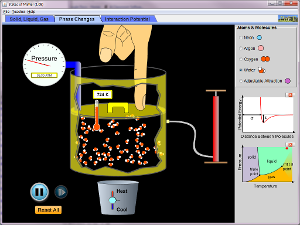
States of Matter Simulation
|
You can download the simulation here. You must have Java Run Time to run this application.
|
|||||||
Atoms, Ions, and Molecules
Chemistry is concerned with the properties of materials. The materials in our world exhibit a striking and seemingly infinite variety of properties, including different colors, textures, solubilities, and chemical reactivities. When we see that diamonds are transparent and hard, table salt is brittle and dissolves in water, gold conducts electricity and can be hammered into thin sheets, and nitroglycerin is explosive, we are making observations in the macroscopic world, the world of our everyday senses. In chemistry we seek to understand and explain these properties in the submicroscopic world, the world of atoms and molecules.
The diversity of chemical behavior results from only about 100 different elements and, thus, only 100 different kinds of atoms. In a sense, the atoms are like the 26 letters of the alphabet that join together in different combinations to form the immense number of words in our language. But how do the atoms combine with each other? What rules govern the ways in which they can combine? How do the properties of a substance relate to the kinds of atoms it contains? Indeed, what is an atom like, and what makes the atoms of one element different from those of another?
The microscopic view of matter forms the basis for understanding why elements and compounds react in the ways they do and why they exhibit specific physical and chemical properties.
The diversity of chemical behavior results from only about 100 different elements and, thus, only 100 different kinds of atoms. In a sense, the atoms are like the 26 letters of the alphabet that join together in different combinations to form the immense number of words in our language. But how do the atoms combine with each other? What rules govern the ways in which they can combine? How do the properties of a substance relate to the kinds of atoms it contains? Indeed, what is an atom like, and what makes the atoms of one element different from those of another?
The microscopic view of matter forms the basis for understanding why elements and compounds react in the ways they do and why they exhibit specific physical and chemical properties.
Atoms
Image credit: Fotolia
An atom is an extremely small particle of matter that retains its identity during chemical reactions. During the latter nineteenth century a series of experiments showed that atoms are comprised of smaller particles. An atom consists of a nucleus and one or more electrons surrounding the nucleus. The nucleus, the core of the atom, has the majority of the mass of the atom, and a positive charge. An electron is a very light particle which circles the nucleus. It has a negative charge. In an electrically neutral atom, the number of electrons equals the positive charge on the nucleus. The nucleus of the atom is composed of two smaller particles called neutrons and protons. A proton has a positive charge equal in magnitude to the negative charge of an electron. This means that in an electrically neutral atom (one with an equal number of protons and electrons), the postive charge of the protons, combined with the negative charge of the electrons, would result in no charge because they would cancel each other out. A proton's mass, however, is a whopping 1836 times that of the electron. A neutron has a mass almost identical to a proton's, but it has no electrical charge associated with it.
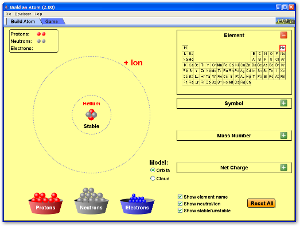
Build an Atom Simulation
|
You can download the simulation here. You must have Java Run Time to run this application.
|
|||||||
Ions
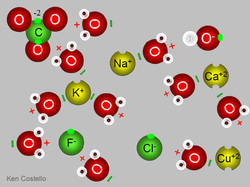
Ions
An ion is an electrically charged particle obtained from an atom or chemically bonded group of atoms by adding or removing electrons. Now what this means is that an ion is the result of taking away, or adding, electrons to an atom or a chemically bonded group of atoms. By taking away, or adding, these electrons, the particle takes on an electrical charge. Atoms are electrically neutral as they contain an equal number of positive and negative charges. An atom that adds an extra electron to it becomes a negatively charged ion. This type of ion is called an anion. An atom which loses one or more of its electrons now has a positive charge, and is called a cation. For example, a sodium atom can lose one of its electrons and form a sodium cation. Now, instead of being Na, it would be Na+1. This means that the sodium atom has an overall positive charge of +1. Another example would be a neutral atom of Sulfur, S. If this atom of S were to gain two electrons it would become S-2. The sulfur atom would now have a total negative charge of -2.
Molecules
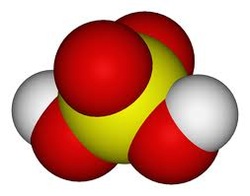
Sulfuric Acid molecule
A molecule is a definite group of atoms that are chemically bonded together. They are tightly connected by attractive forces. A molecular formula is a chemical formula that gives the exact number of different types of atoms in a molecule. Some simple molecular substances are carbon dioxide, CO2; ammonia, NH3; and water, H2O. The atoms that are in a molecule are not just stuffed together without any order. The atoms are chemically bonded to one another in order to form a definite arrangement. A structural formula is a chemical formula which shows how the atoms are bonded to one another to form a molecule. A good example is the structural formula for water, H-O-H. Those two horizontal lines connecting the H with the O (hydrogen and oxygen) represent the chemical bonds joining the atoms.
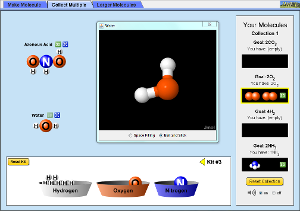
Build a Molecule Simulation
|
You can download the simulation here. You must have Java Run Time to run this application.
|
|||||||
Compounds, Elements, and Mixtures
John Dalton (1766-1844) provided us with the Atomic Theory. His theory says that all matter is composed of small particles called atoms.
- All matter is composed of indivisible atoms. An atom is an extremely small particle of matter that retains its identity during chemical reactions.
- An element is a type of matter composed of only one kind of atom, each atom of a given kind having the same properties.
- A compound is a type of matter composed of atoms of two or more elements chemically combined in fixed proportions.
Compounds
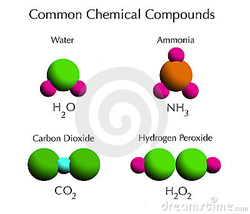
A chemical compound is a pure chemical substance consisting of two or more different chemical elements that can be separated into simpler substances by chemical reactions.Chemical compounds have a unique and defined chemical structure; they consist of a fixed ratio of atoms that are held together in a defined spatial arrangement by chemical bonds. Chemical compounds can be molecular compounds held together by covalent bonds, salts held together by ionic bonds, intermetallic compounds held together by metallic bonds, or complexes held together by coordinate covalent bonds.
Elements

The Periodic Table of Elements
A chemical element is a pure chemical substance consisting of one type of atom distinguished by its atomic number, which is the number of protons in its nucleus. They are divided into metals and non-metals. Familiar examples of elements include carbon, oxygen (non-metals) together with aluminium,iron, copper, gold, mercury, and lead (metals).
As of November 2011, 118 elements have been identified, the latest being ununseptium in 2010. Of the 118 known elements, only the first 98 are known to occur naturally on Earth; 80 of them are stable, while the others are radioactive, decaying into lighter elements over various timescales from fractions of a second to billions of years. Those elements that do not occur naturally on Earth have been produced artificially as the synthetic products of man-made nuclear reactions.
As of November 2011, 118 elements have been identified, the latest being ununseptium in 2010. Of the 118 known elements, only the first 98 are known to occur naturally on Earth; 80 of them are stable, while the others are radioactive, decaying into lighter elements over various timescales from fractions of a second to billions of years. Those elements that do not occur naturally on Earth have been produced artificially as the synthetic products of man-made nuclear reactions.
Mixtures

A yummy mixture!
A mixture is a material that can be separated by physical means into two or more substances. A classic example of a mixture lab would be one in which you were presented a mixture of sand, iron filings, and salt. You are told to separate these materials. How do you do that? Well, think about the various physical properties of each material. You use a magnet to separate out the iron filings. You then mix water with the sand and salt mixture. You swish the water, salt and sand around for a while and then filter it. The salt dissolved into the water, so the salt water solution passes through the filter while the sand gets left in the filter. Now we slowly heat up the salt water solution, and evaporate the water, and we are left with salt. In a mixture, the compounds are not in a definite proportion. For example, a teaspoon of salt in a liter of water is salt water, but so is a cup of salt in a liter of water.
Physical and Chemical Change
Physical Change

Melting ice. Solid to liquid.
A physical change is a change in the form of matter but not in its identity. An example of a physical change would be the dissolving of one thing into another thing. For instance, dissolving sugar into water. The water and the sugar retain their chemical identities and can be separated by physical means. Another example is ice melting to water. Ice and water are both H2O. The identity of the matter is not changed, just the state that it is in.
Chemical Change

Burning wood
A chemical change is a change in which one kind of matter is changed into a different type of matter. Some examples of chemical changes: the rusting of your car, setting your shoe on fire, digesting food, and the burning of magnesium metal in oxygen to form magnesium oxide. All of these materials combine chemically with another material , and cannot be separated by any physical means.