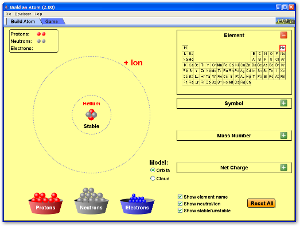
Basic Structure of an Atom

This picture is an example of the arrangement of the particles in an atom. Most of the atom is just empty space. The rest of the atom consists of a positively charged nucleus of protons and neutrons that are surrounded by a cloud of negatively charged electrons. The nucleus is the center of the atom. An atom is an extremely small particle of matter that retains its identity during chemical reactions.During the latter nineteenth century a series of experiments showed that atoms are comprised of smaller particles. An atom consists of a nucleus and one or more electrons surrounding the nucleus. The nucleus, the core of the atom, has the majority of the mass of the atom and a positive charge. An electron is a very light particle which circles the nucleus. It has a negative charge.
In an electrically neutral atom, the number of electrons equals the positive charge on the nucleus. The nucleus of the atom is composed of smaller particles called neutrons and protons. A proton has a positive charge equal in magnitude to the negative charge of an electron. This means that in an electrically neutral atom, the postive of charge the protons, combined with the negative charge of the electrons, would result in no charge because they would cancel each other out. A proton's mass, however, is a whopping 1836 times that of the electron. A neutron, however, has a mass almost identical to a proton's, but it has no electrical charge associated with it.
ParticleProton
Neutron
Electron
|
LocationNucleus
Nucleus
Electron Cloud
|
Weight1.0073 amu
1.0087 amu
0.000549 amu
|
ChargePositive
Neutral
Negative
|
Atomic Number and Mass Number
The atomic number of an element is what distinguishes it from all other elements. An atom's atomic number is the number of protons there are in the nucleus. Hydrogen's atomic number is 1. Helium's atomic number is 2. Any atom that has an atomic number of 1 is a hydrogen atom no matter how many electrons or neutrons the atom has.
The mass number is the number of neutrons added to the number of protons. The mass number of the most common isotope can be obtained from the periodic table. If you take the decimal number on the periodic table and round it to the nearest whole number, you have the mass number. For example the atomic weight of Iron(Fe) is 55.847. When rounded it gives a mass number of 56.
The atomic number of Fe is 26. so most Fe atoms have 30 (56-26) neutrons. In addition, all neutral Fe atoms have 26 protons and 26 electrons. Atoms of the same element with a different number of neutrons are called isotopes. The most common isotope of an element is the one that is on the periodic table.
The mass number is the number of neutrons added to the number of protons. The mass number of the most common isotope can be obtained from the periodic table. If you take the decimal number on the periodic table and round it to the nearest whole number, you have the mass number. For example the atomic weight of Iron(Fe) is 55.847. When rounded it gives a mass number of 56.
The atomic number of Fe is 26. so most Fe atoms have 30 (56-26) neutrons. In addition, all neutral Fe atoms have 26 protons and 26 electrons. Atoms of the same element with a different number of neutrons are called isotopes. The most common isotope of an element is the one that is on the periodic table.
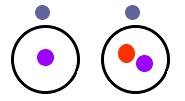
This graphic shows two isotopes of Hydrogen. The picture on the left is the most common isotope of hydrogen with one electron and one proton. The picture on the right is another isotope of hydrogen with one proton, one electron, and a neutron. The most common isotope of uranium is uranium-238 which has 92 protons, 92 electrons, and 146 neutrons. Another isotope is uranium-235 with 92 protons, 92 electrons, and 143 neutrons.
Build an Atom Simulation
|
You can download the simulation here. You must have Java Run Time to run this application.
|
Periodic Table of Elements
The periodic table is a tabular display of the chemical elements, organized on the basis of their atomic numbers and chemical properties. Elements are presented in increasing atomic number. The main body of the table is a 18 × 7 grid, and elements with the same number of valence electrons are kept together in groups, such as the halogens and the noble gases. Due to this, there are gaps that form four distinct rectangular areas or blocks. The f-block is not included in the main table, but rather is usually floated below, as an inline f-block would make the table impractically wide. Using periodic trends, the periodic table can help predict the properties of various elements and the relations between properties. As a result, it provides a useful framework for analyzing chemical behavior, and is widely used in chemistry and other sciences.
Although precursors exist, the current table is generally credited to Dmitri Mendeleev, who developed it in 1869 to illustrate periodic trends in the properties of the then-known elements; the layout has been refined and extended as new elements have been discovered and new theoretical models developed to explain chemical behavior. Mendeleev's presentation also predicted some properties of then-unknown elements expected to fill gaps in his arrangement; most of these predictions were proved correct when those elements were discovered and found to have properties close to the predictions.
All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been synthesized. Of these, all up to and including californium exist naturally; the rest have only been artificially synthesised in laboratories, along with numerous synthetic radionuclides of naturally occurring elements. Production of elements beyond ununoctium is being pursued, with the question of how the periodic table may need to be modified to accommodate these elements being a matter of ongoing debate.
All elements from atomic numbers 1 (hydrogen) to 118 (ununoctium) have been synthesized. Of these, all up to and including californium exist naturally; the rest have only been artificially synthesised in laboratories, along with numerous synthetic radionuclides of naturally occurring elements. Production of elements beyond ununoctium is being pursued, with the question of how the periodic table may need to be modified to accommodate these elements being a matter of ongoing debate.
Avogadro's Number
Avogadro's number and the mole are very important to the understanding of atomic structure. The Mole is like a dozen. You can have a dozen guitars, a dozen roosters, or a dozen rocks. If you have 12 of anything then you would have what we call a dozen. The concept of the mole is just like the concept of a dozen. You can have a mole of anything. The number associated with a mole is Avogadro's number. Avogadro's number is 602,000,000,000,000,000,000,000 (6.02 x 1023). A mole of marbles would spread over the surface of the earth, and produce a layer about 50 miles thick. A mole of sand, spread over the United States, would produce a layer 3 inches deep. A mole of dollars could not be spent at the rate of a billion dollars a day over a trillion years. This shows you just how big a mole is. This number is so large that it is usually only represented in scientific notation.
Probably the only thing you will ever have a mole of is atoms or molecules. One mole of magnesium atoms (6.02 x 1023 magnesium atoms) weigh 24.3 grams. 6.02 x 1023 carbon atoms weigh a total of 12.0 grams. 6.02 x 1023 molecules of CO2 gas only weigh a total of 44.0 grams. The decimal number on the periodic table is the atomic mass, the mass of one atom measured in atomic mass units(amu). Amu's are defined to be 1/12 the weight of the most common isotope of Carbon. This number in grams is the mass of 1 mole of that element. For example, 6.02 x 1023 iron atoms weigh only 55.847 grams.(This is equivalent to saying one mole of iron atoms weighs 55.847 grams.) One mole of sulfur weighs 32.066 grams. (This is the same as saying 6.02 x 1023 Sulfur atoms weigh 32.066 grams)
When not measured in grams, the decimal number on the periodic table is called the atomic mass and is in atomic mass units(amu). As mentioned earlier, one proton weighs 1.0073 amu and 1 neutron weighs 1.0087 amu. So the atomic mass is the mass in amus of one atom of an element, but you rarely use the mass of one atom. Even if you have a tiny speck of a metal or a microgram of an element, you have billions and billions of atoms. Thus, the mass in grams of one mole of an element (the gram atomic weight) is more useful.
Probably the only thing you will ever have a mole of is atoms or molecules. One mole of magnesium atoms (6.02 x 1023 magnesium atoms) weigh 24.3 grams. 6.02 x 1023 carbon atoms weigh a total of 12.0 grams. 6.02 x 1023 molecules of CO2 gas only weigh a total of 44.0 grams. The decimal number on the periodic table is the atomic mass, the mass of one atom measured in atomic mass units(amu). Amu's are defined to be 1/12 the weight of the most common isotope of Carbon. This number in grams is the mass of 1 mole of that element. For example, 6.02 x 1023 iron atoms weigh only 55.847 grams.(This is equivalent to saying one mole of iron atoms weighs 55.847 grams.) One mole of sulfur weighs 32.066 grams. (This is the same as saying 6.02 x 1023 Sulfur atoms weigh 32.066 grams)
When not measured in grams, the decimal number on the periodic table is called the atomic mass and is in atomic mass units(amu). As mentioned earlier, one proton weighs 1.0073 amu and 1 neutron weighs 1.0087 amu. So the atomic mass is the mass in amus of one atom of an element, but you rarely use the mass of one atom. Even if you have a tiny speck of a metal or a microgram of an element, you have billions and billions of atoms. Thus, the mass in grams of one mole of an element (the gram atomic weight) is more useful.
Want more?
Download a lists of documents and tutorials of the related topics. Click the files below.
|
|||||||