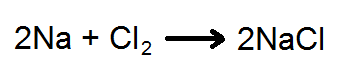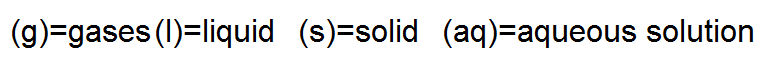What are Chemical Equations?
A chemical equation is the symbolic representation of a chemical reaction where the reactant entities are given on the left hand side and the product entities on the right hand side. The coefficients next to the symbols and formulae of entities are the absolute values of the stoichiometric numbers. The first chemical equation was diagrammed by Jean Beguin in 1615. An example of a chemical equation would be:
This chemical equation would stand for the burning of sodium in chlorine to produce sodium chloride. The formulas on the left side of the equation would stand for the reactant. A reactant is the starting substance in a compound. The arrow, ->, means either "yields" or "reacts to form." The formulas to the right of the arrow stand for the product formed in the chemical reaction. A product is the substance that results from a chemical reaction. The coefficient in front of the Na, 2, give the number of molecules or formula units involved in the reaction. Coefficients of one are usually understood, so they are not written.
Sometimes it helps to indicate the states or phases of the substances in a chemical reaction. We can do this by placing certain labels, which stand for the various phases, following the formula of a substance in a chemical equation. Here are the labels:
Sometimes it helps to indicate the states or phases of the substances in a chemical reaction. We can do this by placing certain labels, which stand for the various phases, following the formula of a substance in a chemical equation. Here are the labels: